Semaglutide for obesity – how does it work, what’s the evidence and can my patient have it?
NO
PHARMACEUTICAL INFLUENCE
Semaglutide for obesity – how does it work, what’s the evidence and can my patient have it?
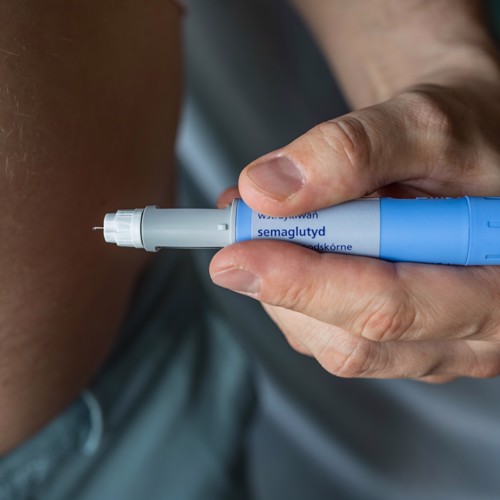
If you have been reading or watching any media recently, you will almost certainly have seen discussion around the new “celeb weight loss drug” or “life changing medication for obesity”, depending upon the views of the editor. Patients are already asking if they can start it, so in this blog I will explain how it works and what it means for those living with obesity.
Wegovy is the brand name for a once a week, injectable version of semaglutide produced by Novo Nordisk for the treatment of obesity. It is used at a higher dose than Ozempic, the brand we are used to using for type 2 diabetes. Semaglutide is a glucagon-like peptide 1 (GLP1) analogue which is a synthetic version of a gut-brain axis hormone initially called “incretin”, because it increased insulin secretion.
GLP1 analogues work on the gut-brain hormone axis, the powerful mechanism that controls our hunger and very importantly our satiety, sending messages to our brain that we are full, making us less hungry and thereby leading to less food being consumed.
In the key STEP 1 Trial, NEJM 2021, in patients with a BMI > 30 semaglutide lead to an average weight loss of 15% of bodyweight over two years compared to 2.4% in the placebo group. Both groups also received a lifestyle intervention. 69% of the semaglutide group had weight loss of > 10%, compared to 12% in the placebo group. There has never been any medication that has been this effective, and the benefits are comparable to metabolic (bariatric) surgery, which remains the best evidence-based weight loss method. In the trial nausea and diarrhoea were the most common side effects, but they were typically mild to moderate, transient and subsided over time.
At the moment, patients can only receive Semaglutide on the NHS via Tier 3 weight loss clinics, and then only for 2 years. It cannot be prescribed to patients by GPs, except in the form of Ozempic for patients with Type 2 diabetes. If your patients would benefit from weight loss alongside their Type 2 diabetes, then using a GLP1 analogue would be a sensible decision and is supported by the new NICE guidelines.
As soon as Semaglutide is stopped, the patient will likely begin to regain weight, as happens with almost all weight loss methods including food restriction. This is due to the effects of the gut-brain axis changing the hormone balance to regain weight. It is NOT due to people with obesity “not having enough willpower”.
This leads to an interesting ethical debate around the decision to limit the prescribing of the drug to two years. Would we limit antihypertensives to 2 years, or asthma meds? We accept that these are chronic diseases that need lifelong treatment. So why not obesity, which also meets the criteria for a life-long relapsing remitting chronic disease, and is accepted as such in many countries? Is it acceptable to prescribe medication for a limited time, knowing that the condition will return through no fault of the patient?
The reason is obesity stigma.
There is still a widespread belief in both society, and the medical world, that people with obesity have brought it upon themselves and just need to try harder to lose weight. This leads to the conclusion that using a drug to lose weight is somehow cheating, or taking the easy way out, and we shouldn’t be spending NHS money on something people could “fix” themselves.
This argument is incorrect and treating obesity properly, would prevent spending money on the complications of obesity that currently cost the NHS £6 billion per year. But it will require a huge shift in mindset of society and those with the decision-making power for NHS budgets.
Obesity is sadly misunderstood by many health professionals, and an important educational need. If this blog has interested you and you would like to know more about the causes of obesity and how we can help our patients living with it better, please join me and Neal Tucker on Thursday March 9th for our latest NB Medical Obesity Webinar.
Dr Stephanie De Giorgio
2nd March 2023
Join us at one of our upcoming courses or live webinars

Recent NB Blogs
Need an immediate update? – all our courses are available on demand
Did you find this useful?
You can quickly add CPD to your account by writing a reflective note about the Semaglutide for obesity – how does it work, what’s the evidence and can my patient have it? post you've read.
Log in to your NB Dashboard and use the 'Add Reflective Note' button at the bottom of a blog entry to add your note.

